
 | Home |
 | Properties |
|  | Pure Component |
| |  | Water |
| |  | Physical |
| | |  | Rules of Thumb |
| | |  | Common Relationships |
| | |  | Chem & Physical Data |
| | |  | Physical Properties Data |
| | |  | Density |
| | |  | Boiling Point |
| | |  | Melting Point |
| | |  | Vapor Pressure |
| | |  | Surface Tension |
| | |  | Thermal Expansion |
| | | |  | Liquids |
| | |  | Critical Constants |
| | | |  | Critical Temperatue |
| | | |  | Critical Pressure |
| | | |  | Critical Volume |
| | | |  | Critical Compressibility |
| |  | Thermodynamic |
| | |  | Enthalpy |
| | |  | Entropy |
| | |  | Heat Capacity |
| | |  | Heat of Vaporization |
| | |  | Heat of Fusion |
| | |  | Heat of Formation |
| | |  | Free Energy of Formation |
| | |  | Heat of Combustion |
| | |  | Fugacity |
| | |  | Explosive Limit and Flash Point |
| |  | Molecular Transport |
| | |  | Thermal Conductivity (thermal diffusivity) |
| | |  | Mass Diffusivity |
| | |  | Viscosity (momentum diffusivity) |
|  | Multi-Component |
| |  | Air-Water |
| | |  | Relative Humidity |
| |  | Aqueous HCl |
| |  | Relative Volatility |
| |  | Physical Properties |
| | |  | Physical Properties of Mixtures |
| | |  | Bubble Point & Dew Point |
| | |  | Solubility |
| | | |  | Water |
| | | |  | Solvent |
| |  | Thermodynamic |
| | |  | Thermodynamic Properties of Mixtures |
| | |  | Activity Coefficients |
| | |  | Explosive Limit & Flash Point |
| |  | Molecular Transport Properties |
| | |  | Molecular Transport Properites of Mixtures |
 | Safety |
|  | Explosive Limit and Flash Point |
|  | Inerting |
|  | Ventillation |
|  | Pressure Relief |
| |  | Relief Valves |
| |  | Rupture Discs |
| |  | Explosion Panels |
 | Unit Operations |
|  | Fluid Mechanics |
| |  | Statics |
| |  | Dynamics |
| | |  | Liquid Agitation |
| | |  | Pumps & Compressors |
| | |  | Vacuum Systems |
| | |  | Line Loss |
| | |  | Pipe Equivalent Lengths |
| | |  | Affinity Laws for Pumps & Fans |
| | |  | Fixed-Bed Pressure Drop |
| | |  | Orifice Plates |
| | |  | Control Valves |
| | |  | Flow Meters |
| | |  | Open Channel Flow |
|  | Heat Transfer |
| |  | Conduction |
| |  | Convection |
| | |  | Forced Convection |
| | | |  | Heat Transfer Coefficients |
| | | | |  | Film Coefficients |
| | | | |  | Agitated Tank Heat Transfer |
| | | | |  | Overall Coefficients |
| | | |  | LMTD Calculations |
| | | |  | Batch Heat Transfer Time |
| | | |  | Shell & Tube Exchangers |
| | | |  | Nucleate Boiling & Reboilers |
| | |  | Natural Convection |
| |  | Radiation |
|  | Mass Transfer |
| |  | Diffusion |
| | |  | Diffusion thru Stagnant Gas Film |
| | |  | Vaporization Rate of Liquid |
| |  | Distillation |
| | |  | Batch |
| | |  | Batch-Column |
| | |  | Continuous Equilibrium Stages |
| | |  | Continuous Contacting |
| |  | Absorption & Stripping |
| |  | Flash Calculations |
| |  | Extraction |
| |  | Leaching |
|  | Combined Heat & Mass Transfer |
| |  | Drying |
| |  | Evaporation |
| |  | Humidification |
|  | Chemical Kinetics |
|  | Separation Processes |
| |  | Filtration |
| |  | Centrifugation |
| |  | Sedimentation |
| |  | Crystallization |
| |  | Screening |
 | Special Processes |
|  | Steam Reforming |
 | Thermodynamics |
|  | State Functions |
| |  | Mechanical Properties |
| | |  | Pressure (p) - Intensive |
| | |  | Volume (V) - Extensive |
| |  | Fundamental Properties |
| | |  | Temperature (T) - Intensive |
| | |  | Internal Energy (E) - Extensive |
| | |  | Entropy (S) - Extensive |
|  | Path Functions |
| |  | Work (W) |
| |  | Heat (Q) |
|  | Composite Properties |
| |  | Enthalpy (H) |
| |  | Work Function (A) |
| |  | Gibbs Free Energy (G) |
 | Chemistry |
|  | Rules of Thumb |
|  | Periodic Table |
|  | Thermochemistry |
| |  | Heat of Formation |
| |  | Heat of Combustion |
| |  | Heat of Reaction |
|  | Reaction |
| |  | Equilibrium |
 | Tools |
|  | Math |
| |  | Linear Regression |
|  | Conversions |
 | Help and Tips |
 | Videos |
 | Site Map |
|
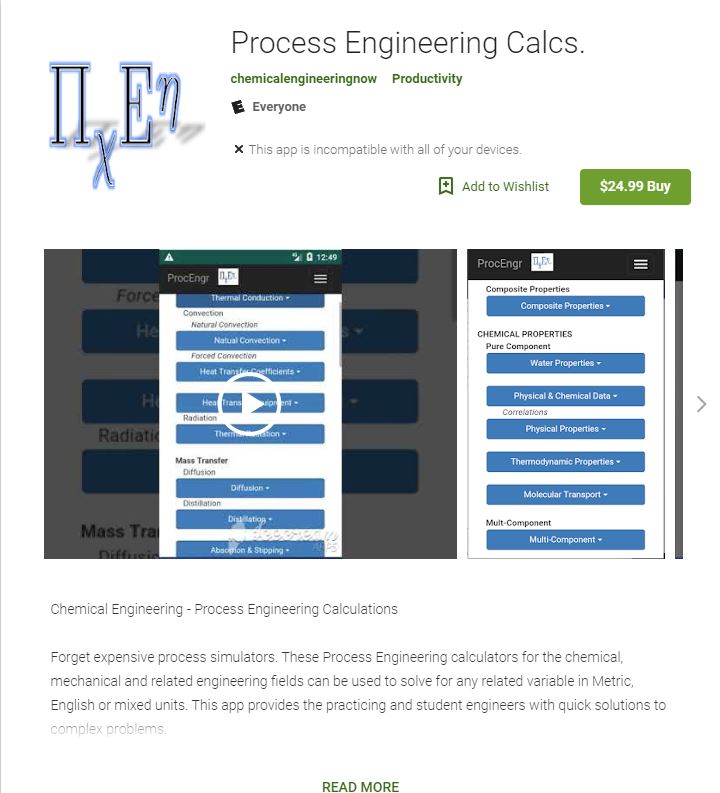
We are now available in the Google Play Store for Android devices as “Process
Engineering Calcs”. Not yet available for Apple iPhone, but coming soon.
Click here to go to Google Play Store.
May 1, 2019 – Over the next two weeks, this site will remain open for your
benefit. After which, the site resources will be redirected in full support of
the new Process Engineering Calcs. App.
We have made a big investment into this new application in order for it to be
more useful, mobile, and of greater value to you. You will want to keep this App
at your side at all times.
Please go to the Google Play Store and download your copy; and please remember
to rate the app in the Play Store after using it.
We want to thank all of our followers from over the years and hope that each and
everybody received great benefit from ChemicalEngineeringNow. We look forward to
continue serving you with Process Engineering Calcs as we will be continuing to
improve and expand functionality.
Also, please be so kind as to share this information with your friends and
colleagues.
|
|